Co-authors Matt Gibbs CipherBio@svb and Katherine Andersen @katherun Head of SVB Life Science and Healthcare Banking.

If the coronavirus has proven anything, it is that size and impact are not necessarily correlated. A 125-nanometer parasite that cannot be seen with the naked eye, the COVID-19 virus has killed over 160,000 people globally and infected 2.4M people worldwide. It has exposed systemic vulnerabilities in national healthcare systems and marked the emergence of new global trends. In the life sciences sector, it merely reinforced an already existing trend characterized by increasing collaboration and more projects that rely on the collective contributions of various partners. Today, the sector is proving to be highly agile and primed for impact. Collaborations in the sector are taking place across countries, rival companies, and public and private organizations, with the single aim of putting an end to the COVID-19 pandemic.
VC backed startups and established companies fighting the pandemic
Venture backed private companies and public companies are fighting the virus from different angles. They are aiming to provide effective diagnostic tools, vaccines, and treatments to deal with the virus for the benefit of patients and exposed healthcare staff. Below, a few startups in addition to more established companies have been highlighted.
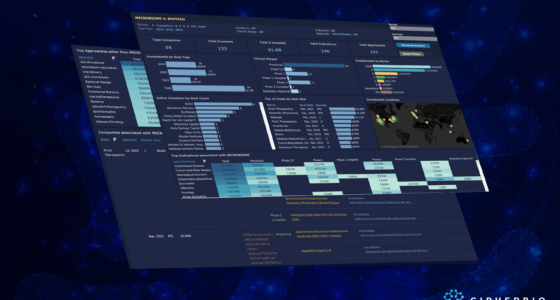
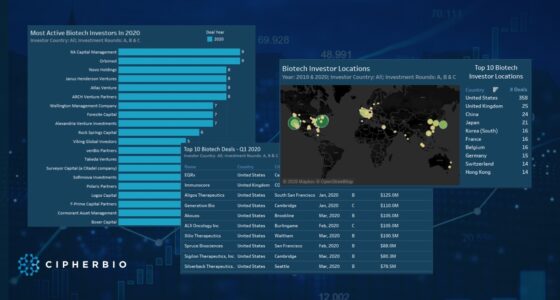
 CipherBio Insights: Life Science companies and investors actively responding to COVID-19.
CipherBio Insights: Life Science companies and investors actively responding to COVID-19.
Companies working on therapeutics and treatments
Vir Biotechnology is manufacturing human monoclonal antibodies for the potential treatment of COVID-19 after it signed a letter of intent with Biogen Inc.
Some AI and digital health companies such as viz.ai, Doctors on Demand, and Ginger.io are offering their platforms to provide telehealth services for patients as well as training for healthcare staff. In 2018, Doctors on Demand had raised $74 million in Series C financing round which was led by Princeville Global and Goldman Sachs Investment. The company has backing also from other investors such as Alta Partners, and SoftBank Vision Fund. Viz.ai had seed and series A funding from Innovation Endeavors, and series B funding from Google Ventures, and Kleiner Perkins.
|
Top Investors in Digital Health/AI companies working on COVID-19. |
Companies working on coronavirus vaccines

Moderna (MRNA) has been among the early companies to work on a vaccine. The company was co-founded by Bob Langer of Langer Labs MIT and Noubar Afeyan, Ph.D, who is the CEO of Flagship Pioneering. Moderna marked the biggest biotech IPO in history raising $604.3 million in 2018. In April 2020 Moderna received a $483M investment from BARDA to speed the development of Moderna’s Coronavirus vaccine, driving the Moderna market cap to $15B. Viking Global Investors, Abu Dhabi Investment Authority, and EDBI are among the investors in ModernaTX. The company managed to finish the sequence for its vaccine (mRNA-1273) only two days after it learned about the genetic sequence of the virus, and finished the first clinical batch within less than a month from that date. Currently, the Emory University in Atlanta is testing the vaccine on adult volunteers in its first phase study. Moderna designed the vaccine in collaboration with the Vaccine Research Center (VRC) of the National Institute of Allergy and Infectious Diseases (NIAID), part of NIH. The manufacturing of the virus is funded by The Coalition for Epidemic Preparedness Innovations (CEPI). Moderna has a good track record when it comes to previous clinical trials of its vaccines.

Sorrento Therapeutics (SRNE) is also working on a COVID-19 vaccine called STI-6991 and is currently getting guidance from the FDA on its development. The vaccine will enter into clinical trials potentially in the second half of 2020. If approved, the company is expected to have the capacity to manufacture the vaccine on a global scale.

Versatope Therapeutics, which was selected as a resident at JLABS incubator, has chosen the universal influenza vaccine for clinical development to use against the COVID-19. Versatope’s approach combines diverse variants of influenza strains on a single nano-sized recombinant outer membrane vesicles (rOMVs) that may provide better cross-strain protection than influenza vaccines composed of individual strains.

Twist Bioscience (TWST), a VC backed San Francisco-based synthetic biology company, secured $26 million in series B financing in 2014. The round was led by Tao Invest, with participation from ARCH Venture Partners, Paladin Capital Group, and other investors. The company secured $61M in series D funding from investors such as Biomatics Capital, Bay City Capital, FIS, and Ditch Plains Capital Management and $70M in an IPO in 2018. Currently, the company is offering genomic tools to classify and characterize viral samples, as well as genomic engineering tools to develop a vaccine. The company’s core offering is a silicon-based DNA writing platform that allows for the rapid development of Next-generation sequencing (NGS) panels.
|
Investors in companies working on the COVID-19 vaccine. |
CuraVac AG, a Germany-based biopharmaceutical company in the clinical stage is working on developing a vaccine against the virus based on the mRNA molecule.
Inovio Pharmaceuticals, (INO) initiated the first phase of the clinical trial of its vaccine on 6 April.

Janssen Pharmaceutical, a subsidiary of Johnson & Johnson, struck a partnership agreement with the Beth Israel Deaconess Medical Center to develop a COVID-19 vaccine. The partners are currently working on preclinical trials to select vaccine candidates. Jenseen is using its AdVac and PER.C6 technologies in this project.
Novavax (NVAX) a publicly traded biotech company on the Nasdaq, plans to initiate clinical trials of its coronavirus vaccines in May. The vaccine is named NVX-CoV2373 (after a stable perfusion protein) and is made using the nanoparticle technology of the company. The vaccine showed promising results in animal studies.
Tonix Pharmaceuticals is also working on a vaccine but taking a different approach, as they are using a live replicating horsepox virus as a vector for antigens from the SARS-CoV-2 virus. The replicating virus is supposed to induce cellular immunity instead of humoral immunity or antibodies. Despite this, the company is attempting to induce both types of immunity responses rather than only one.

BioNTech is a germany-based biotech company with backing from VCs such as Fidelity Management and Research Company, Redmile Group, and Platinum Asset Management. It is currently partnering with Pfizer with the purpose of developing a vaccine against COVID-19. BioNTech would contribute with mRNA vaccine candidates. This is a part of BioNTech’s clinical program called BNT162 which should enter clinical trials in April.
|
VCs investing in BioNTech. |
Companies working on detection, diagnosis, and testing

PathGroup, one of the largest private providers of comprehensive anatomic, clinical and molecular pathology laboratory services in the US, has received the 1000th panther system placement form Hologic. The panther system is a molecular diagnostic testing system that can be employed for the diagnosis of the COVID-19 disease. The company completed recapitalization with Pritzker Group Private Capital in 2016.

Quidel, a US-based manufacturer of cell culture products and a publicly traded company on Nasdaq, is currently offering its quick test that offers results in less than 2 hours to COVID-19 patients. The test relies on extractions from the patient’s Nasopharyngeal (NP) or Oropharyngeal (OP) swab.

E25Bio, a startup that emerged from the Engine at MIT which specializes in producing diagnostics for highly infectious diseases, was able to raise $2 million in funding from Khosla Ventures, to make a diagnostic test for COVID-19.

Inflammatix, which is a part of the StartX task force, had announced a breakthrough in Diagnosing Acute Infections by Reading the Immune System, which could help against the novel virus. The company had raised $32 million in series C funding from Khosla Ventures, Northpond Ventures, Think.Health Ventures and new investor Grey Sky Venture Partners.

Everlywell is a Texas-based startup, The company had raised $50 million in funding last year to accelerate the development of its testing platform. Highland Capital Partners, NextGen Venture Partners, and Next Coast Ventures are among the key VCs backing Everlywell. The company is responding to COVID-19 with its test kit that can be used at home and enables consumers to get telehealth diagnosis for COVID-19. The company prioritized healthcare workers and allocated the first batches of the tests to them. The tests were available to consumers on March 23rd. To scale the production of the test, Everlywell announced a $1 million-dollar development incentive to any certified laboratories who fulfilled requirements set by the FDA and Everlywell to prioritize the development of a COVID-19 diagnostic test.

Mammoth Biosciences has the support of many investors including Mayfield Fund and aMoon fund. The company offers the CRISP-based detection platform which provides molecular-based diagnosis and has prototyped a rapid test kit based on that platform to detect the coronavirus in humans. The expected benefit of this is that it offers speedy and accurate results and is highly scalable. The company had secured $45 million in series B funding earlier this year in a round led by Decheng Capital. Jennifer Doudna, an early gene-editing pioneer, co-founded Mammoth Biosciences.

Ginger.io provides on-demand coaching services for health workers. The company raised $35 million in series C funding round led by WP Global Partners. Kaiser Permanente, Khosla Ventures, and City Light Capital are among the VCs currently backing the company.

OpenTrons, a Brooklyn based company aiming to automate biologists’ work, is currently leveraging its robotic capabilities to test for the coronavirus at scale. Testing vast populations has proven effective in combating the virus in South Korea. The company had raised $10 million in seed funding from Khosla Ventures, Lerer Hippeau Ventures, Y Combinator continuity fund, and Jeffery Kindler.

Oura Health, which makes smart rings that read the body’s vital signs such as the heart and respiratory rates, can also help in early detection of the COVID-19 based on temperature readings. A study is currently being carried out with the participation of 2000 medical workers who would wear the rings for 3 months. Tesi and MSD Capital are among the investors in Oura Health.

Kinnos, a US-based company that focuses on protection against infections, manufactures color additives to disinfectants to help people ensure that surfaces are covered and clean. It has secured $6 million in a funding round in March 2020 led by Prolog Ventures. It has seed backing from VentureWell, Georgica Advisors, and New York Angels.

Avalon Healthcare Solutions has the backing of investors like Francisco Partners and BlueCross BlueShield of South California. It offers a laboratory benefits management platform and has grown exponentially in recent years. It is working on coordinating testing for COVID-19. The company is currently focusing on coordinating testing efforts, and has discussed increasing efficiency of testing with the government. It is also working to provide its clients with access to labs that can perform COVID-19 testing.
|
Investors in companies working on detection, diagnosis, and testing for COVID-19. |
Collective efforts and large scale collaborations in the life science sector to end the pandemic
In addition to the individual companies, there are many examples of collaboration and pooling of resources that can be mentioned. The Life Sciences Consortium (founded in 2005 to improve cooperation between different companies to advance oncology treatments), the Medical Device Innovation Consortium, Transcelerate, and The Coalition for Epidemic Preparedness Innovations (CEPI), are just a few. Currently, the already established collaboration trend is gathering some steam. Various companies, organizations, and experts are participating in collective efforts with improved coordination and synchronization between them. Some are mentioned below.
StartX Med COVID-19 Task Force

StartX is both a non-profit accelerator and a community that includes various actors such as professors from Stanford, experts from the life sciences sector, startups in the growth stage, and entrepreneurs. It was established in 2011 and today it contains more than 700 startup companies, each having raised around $10 million on average. DawnLight Technologies, Branch Metrics, freenome, and Periscope are just a few examples. In March, StartX launched a task force that includes experts from 45 companies to work on the prevention, diagnosis, and treatment of the COVID-19.
C3.ai digital transformation institute

The founder of C3.ai, an artificial intelligence company, has initiated a consortium that includes public and private institutions such as biotech companies and universities to fight the virus. The consortium is called C3.ai digital transformation institute, and it pledged to spend $367 million in the upcoming five years, giving priority to speedy solutions. Microsoft, MIT, and Princeton University are but a few names in the consortium, which has the purpose of establishing the new Science of Digital Transformation of Societal Systems. Currently, the Consortium is using its resources to address the COVID0-19 pandemic. The institute called for research proposals on “AI techniques to mitigate pandemic” from scholars, developers, and researchers.
World Health Organization (WHO) launches SOLIDARITY trial

The WHO launched in late March a large trial of a selection of drugs based on factors like success probability, potential hazards, and availability of supplies. The SOLIDARITY research will test the already approved drugs which are remdesivir (an antiviral); the malaria medications chloroquine and hydroxychloroquine; a combination lopinavir and ritonavir (of two HIV drugs); and that same combination plus interferon-beta, an immune system messenger that can help against viruses.
The European Union launched clinical trials under the name “DISCOVERY”
In late March, the EU launched clinical trials for various COVID-19 treatments. The study would include approximately 3200 patients from 🇫🇷France, 🇧🇪Belgium, 🇩🇪Germany, 🇱🇺Luxembourg,🇳🇱Netherlands, 🇪🇸Spain, 🇸🇪Sweden, and the 🇬🇧United Kingdom. It is coordinated by Inserm (The French National Institute of Health and Medical Research), which is a part of the reacting consortium. The “DISCOVERY” trial will test the same drugs used in SOLIDARITY trial except for Chloroquine. The EU has already allocated €140 million for research towards a cure and vaccine.
 To overcome some challenges related to the testing, a team from JRC labs in Belgium developed a new control material to ensure accurate results and facilitate quality control. The new material will likely increase the efficiency of testing and improve the allocation of resources to that end.
To overcome some challenges related to the testing, a team from JRC labs in Belgium developed a new control material to ensure accurate results and facilitate quality control. The new material will likely increase the efficiency of testing and improve the allocation of resources to that end.
The European Commission is also providing other forms of support. It has postponed regulation on medical devices for at least one year to remove hurdles in the way of companies that can help. It has also lifted duties and VAT on needed imports such as medical devices and protective equipment. The move reduces the prices of those products by approximately a third.
Y Combinator companies respond to the pandemic

Y Combinator, a US-based accelerator, has listed the companies in its portfolio that are actively working on tests, diagnostics, or vaccines against COVID-19. To mention a few examples, Unima from Mexico is offering an ultra-low-cost test that uses only a strip and a smartphone, and Gen1 E lifesciences is working on a drug to treat Acute Respiratory Distress Syndrome (ARDS) that can result from the virus infection. Among YC companies working on combating the virus are MacroMoltek, YourChoice Therapeutics, Atomwise, PostEra, and others. Although the efforts between the companies are not coordinated at a higher level, this does show massive mobilization at the sector level towards a solution.
Supercomputers join the fight against coronavirus
In this tireless fight, some efforts have enlisted the help of a new conscript: supercomputers. IBM’s supercomputer, Summit, which was dubbed the world’s fastest computer in November last year, ran numerous simulations to analyze which drug compounds might effectively stop the virus from infecting host cells. The analysis aimed to repurpose molecules based on the similarities between the old SARS-CoV and SARS-CoV2. Summit has thus far identified 77 compounds as potential candidates, and the results have been published in ChemRxiv. This stands to confirm that data is the vital factor that would tip the scale in our favor during this challenge.

Summit and Sierra, supercomputers at Oak Ridge National Laboratory and Lawrence Livermore National Laboratory.
Data on the genetic sequence of the virus

Since the beginning of the outbreak, sharing data about the genetic makeup of the virus has been critical. Various countries are now sharing their findings and thus far the genetic sequences of over 80 COVID-19 strains have been made available at genome data banks, such as the GenBank database at the National Center for Biotechnology Information (NCBI) in the United States. Data is also provided by GISAID which offers public access to genetic sequence data of influenza viruses and other relevant data through its database.
Machine learning is breaking cooperation barriers

Big data, artificial intelligence, and machine learning are proving to be powerful weapons, but that is not only because of their analytical power and problem-solving abilities. It is also because they are removing long-standing barriers in the way of cooperation between different actors. For instance, last year, ten large pharmaceutical companies and seven large tech companies joined forces in a project called Machine Learning Ledger Orchestration for Drug Discovery (Melloddy). The firms would contribute with data, while maintaining their proprietary information, to a single blockchain-based platform to discover new drugs. Federated learning, a machine learning technique introduced by Google AI, enabled those companies to analyze each other’s local data without any exchange of that data between different parties. This quelled IP infringement fears, which had hindered cooperation in the past.
Contributions from Non-life science companies
Other companies from other domains are also adding positive to the global response to the virus. Twitter founder, Jack Dorsey, pledged to donate $1 billion (28% of his wealth) to fund research to help “disarm the pandemic”. Tesla is working on building its own ventilators made out of car parts. Apple and Google are partnering up and adding a technology to their smartphone operating systems to trace whether people have contacted an infected person. Apple had earlier pledged donations of $15 million and is using its abilities to make face shields. Microsoft founder, Bill Gates, who was very prescient about the virus outbreak a few years ago, pledged $100 million for the fight through the Bill and Melinda Gates Foundation. The foundation added another $150 million recently, making its total commitment $250 million.
|
|
Louis Vuitton is using its production lines normally used to make perfumes and make-up to make hand sanitizers. Facebook will be surveying users about COVID-19-related symptoms as part of a Carnegie Mellon University research project to generate a heatmap of self-reported cases. Even talented young men and women are contributing according to their own abilities, as they are designing protective equipment for healthcare workers using 3d printing and building websites that gather the latest statistics about the virus outbreak.
Final words
The theme in the life sciences sector is characterized by collaboration, collation, joint endeavor, and synergy. Economies of scale of data are emerging, and with the help of various technologies, this promises to deliver an unprecedented positive impact. Defeating COVID-19 for good is only the beginning, as now various companies are seeing the extent of the mutual benefits of collaboration. This current theme offers surplus to companies, investors, and individuals, in terms of health, advanced knowledge and practices, and good financial bottom-line. The COVID-19 may have inadvertently kickstarted or accelerated a cooperation that benefits us years to come, and optimism is highly warranted.
The efficiency of the life sciences sector depends on your participation
 We at CipherBio are working to support companies in the life sciences sector and investors in all of their data and information needs. Our strong network and connections with both investors and startups enable us to present you with the most timely, accurate, and comprehensive database about the entire life sciences ecosystem.
We at CipherBio are working to support companies in the life sciences sector and investors in all of their data and information needs. Our strong network and connections with both investors and startups enable us to present you with the most timely, accurate, and comprehensive database about the entire life sciences ecosystem.
Join us in our effort to capture the entire life science ecosystem in our database
Fill out your digital profile, or update it with your company’s latest news, so that you can be a part of the data set that represents the life science ecosystem. |
































 We strongly believe that the availability of a high-quality database about the life sciences ecosystem increases sector efficiency and benefits every company raising capital. Thus, if you are an investor or a startup in any of the relevant industries, we would like to extend
We strongly believe that the availability of a high-quality database about the life sciences ecosystem increases sector efficiency and benefits every company raising capital. Thus, if you are an investor or a startup in any of the relevant industries, we would like to extend